New antiviral treatment for COVID-19 will be mostly set aside for use in older unvaccinated individuals and immunocompromised Ontarians
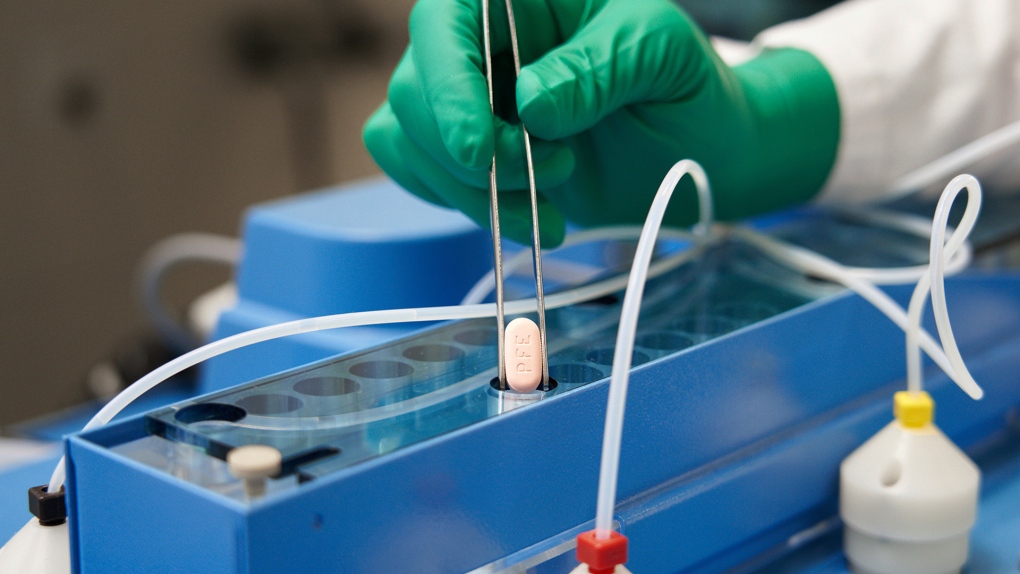 This image provided by Pfizer in October 2021 shows the company's COVID-19 Paxlovid pills. (Pfizer via AP)
This image provided by Pfizer in October 2021 shows the company's COVID-19 Paxlovid pills. (Pfizer via AP)
A new antiviral drug that can be used to treat COVID-19 will be primarily set aside for use in older unvaccinated individuals and immunocompromised Ontarians.
The Ford government has released new eligibility guidelines that provide the first look at how it will distribute its limited supply of Pfizer’s Paxlovid pill, the first shipment of which arrived in the province last week.
The government says that it is prioritizing individuals at “the highest risk of severe outcomes,” including unvaccinated Ontarians age 60 and up and unvaccinated Ontarians aged 50 and up who also have one or more specific risk factors.
The government will also make all immunocompromised individuals aged 18 and up eligible, regardless of their vaccination status, as well as all unvaccinated First Nation, Inuit and Métis individuals aged 50 and up.
In order to access the drug those eligible will have to first test positive for COVID-19 and the Ford government says that it is now introducing “expanded COVID-19 assessment centres” where Ontarians will be able to access both testing and “outpatient therapeutics,” such as Paxlovid.
It says that the testing centres will have access to multiple testing options, including rapid tests, to asses those who may be eligible for treatment.
However, not all assessment centres will have the drug on hand.
According to data from the clinical trials, the Paxlovid pill was found to be 89 per cent effective at reducing the risk of hospitalization and death after a full course of treatment (three pills twice daily for five days in a row).
The Ontario government has said that it expects to receive approximately 10,000 courses of treatment in January.
“Given the very limited supply of antivirals, not all patients who meet the clinical eligibility criteria will be guaranteed access to treatment,” the government warns on a new website set up to provide information about antiviral treatments.
Paxlovid was approved by Health Canada on Jan. 17.
CTVNews.ca Top Stories

'Most of the city is evacuating': Gridlock on Alberta highway after evacuation order in Fort McMurray
Four Fort McMurray neighbourhoods were ordered to evacuate on Tuesday as a wildfire gets closer to the city.
Sask. police seize 1.5M pieces of evidence, lay 60 more charges in child exploitation case
Saskatchewan RCMP have revealed that a historic sexual assault investigation has led to the discovery of alleged crimes against children dating back to 2005.
'Inappropriate' behaviour shuts down Dublin to New York City portal
Less than a week after two public sculptures featuring a livestream between Dublin, Ireland, and New York City debuted, 'inappropriate behaviour' in real-time interactions between people in the two cities has prompted a temporary shutdown.
Oilers starting Calvin Pickard in goal for Game 4 vs. Canucks
The Edmonton Oilers will start Calvin Pickard in net Tuesday for Game 4 of their playoff series with the Vancouver Canucks.
Biden administration moving ahead on US$1 billion arms package for Israel, AP sources say
The Biden administration has told key lawmakers it is sending a new package of more than US$1 billion in arms and ammunition to Israel, two congressional aides said Tuesday.
King Charles III unveils his first official portrait since his coronation
King Charles III has unveiled the first portrait of the monarch completed since he assumed the throne, a vivid image that depicts him in the bright red uniform of the Welsh Guards against a background of similar hues.
Full List Are these Canada's best restaurants? Annual top 100 list revealed
The annual list of Canada's top restaurants in the country was just released and here are the places that made the 2024 cut.
Alberta announces the 4 health agencies that will replace AHS later this year
The province has released more information on its plan to break up Alberta Health Services and replace it with four sector-based health agencies.
Maximum payout for LifeLabs class-action drops from $150 estimate to $7.86
Canadian LifeLabs customers who filed an application for a class-action settlement began receiving their payments this week, though at a much lower amount than initially expected.